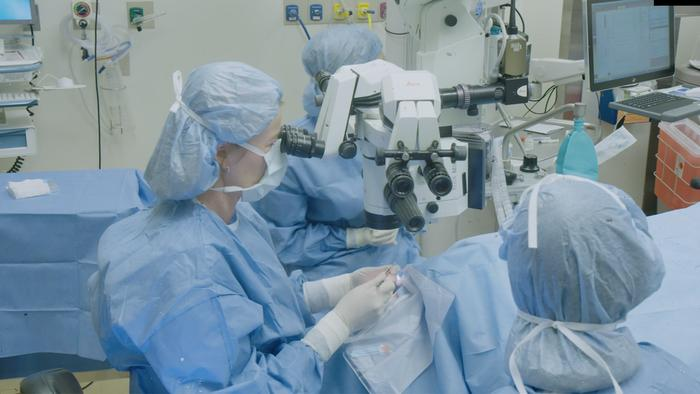
Therapie CALEC, a groundbreaking advancement in ocular medicine, is revolutionizing the treatment of severe corneal damage previously deemed untreatable. Developed by the esteemed team at Mass Eye and Ear, this innovative stem cell therapy harnesses the power of limbal epithelial cells from a healthy eye, facilitating corneal repair in those suffering from ocular injuries. The therapy has demonstrated remarkable efficacy, boasting over a 90% success rate in restoring corneal surfaces among participants in a clinical trial. With its promising results, CALEC offers renewed hope for individuals who endure persistent pain and vision challenges due to corneal deficiencies. As research continues and further trials are conducted, CALEC may soon transform how we approach ocular injuries and corneal restoration, marking a significant step forward in eye care.
The innovative technique known as autologous cultivated limbal epithelial cells (CALEC) represents a significant leap in regenerative ocular therapies, focusing on the restoration of the corneal surface. By utilizing stem cells harvested from a healthy donor eye, this method has shown impressive results in treating ocular traumas that lead to vision impairment. The process not only aims to mend the delicate corneal tissue but also mitigates the chronic discomfort often associated with corneal deficiencies. With backing from reputable institutions like Mass Eye and Ear, CALEC stands at the forefront of therapeutic interventions designed to enhance visual rehabilitation for patients suffering from serious corneal damage. Continued exploration in this field promises to expand treatment options, potentially benefiting those afflicted by corneal injuries.
Introduction to CALEC Therapy
The breakthrough in corneal repair known as CALEC therapy marks a significant advancement in the field of ophthalmology. Developed at Mass Eye and Ear, this innovative procedure utilizes autologous cultivated limbal epithelial cells to restore the cornea’s surface after serious ocular injuries. By taking healthy cells from a donor eye and growing them in a lab, CALEC therapy offers new hope for patients suffering from conditions that were previously thought untreatable.
With the preliminary results showing over 90 percent effectiveness in restoring corneal surface integrity, CALEC therapy stands to redefine treatment protocols for ocular injuries. This significant success rate highlights the potential for stem cell therapy in the modern medical landscape, providing a viable alternative to traditional corneal transplants that often do not yield satisfactory results.
Understanding Limbal Epithelial Cells in Ocular Health
Limbal epithelial cells play a crucial role in maintaining the health of the cornea, the eye’s transparent outer layer. These cells are responsible for regenerating and sustaining the corneal epithelium, ensuring its smooth surface and protective barrier against infections and irritants. When ocular injuries, such as chemical burns or trauma, deplete these vital cells, patients can experience severe pain and loss of vision, underscoring the importance of limbal epithelial cells in ocular health.
Research has shown that when patients suffer from a deficiency of limbal cells, the possibility of successful corneal transplants diminishes. This is where therapies like CALEC become essential. By cultivating these cells, physicians can effectively regenerate the corneal surface, restoring vision and alleviating painful symptoms associated with limbal cell deficiency.
The Groundbreaking Clinical Trials at Mass Eye and Ear
The recent clinical trials at Mass Eye and Ear have provided groundbreaking insights into the efficacy of CALEC therapy. In the study, conducted over 18 months with 14 participants, researchers successfully demonstrated that this method could restore the corneal surface, even after severe damage caused by ocular injuries. The trials, which were rigorously designed and approved by regulatory bodies, emphasize the safety profile of this stem cell therapy.
Participants in the study experienced significant improvements in their vision and quality of life, with a notable percentage achieving complete restoration of corneal integrity. Such outcomes render CALEC therapy not only a scientific breakthrough but also a beacon of hope for countless individuals facing irreversible ocular damage.
Future Directions for Stem Cell Therapy in Ocular Care
The future of stem cell therapy in ocular care is promising, especially with the advancements made through CALEC. Researchers are now exploring the potential for allogenic grafts, which would allow for application in patients with bilateral ocular damage. This shift could significantly broaden the accessibility of CALEC therapy, ensuring that more patients can benefit from its restorative capabilities.
As ongoing studies continue to gather data, including multi-center trials with larger patient populations, the hope is that these findings will drive the FDA toward approving CALEC therapy for clinical use. The urgency for such developments cannot be overstated, as it could profoundly affect the lives of individuals suffering from debilitating eye conditions.
Challenges and Limitations of Current Ocular Treatments
Despite the advances represented by CALEC therapy, challenges remain in the realm of ocular treatments. Current limitations necessitate that patients have only one eye affected in order to harvest the necessary limbal cells from a healthy eye. This restriction can be a significant barrier for many individuals, particularly those with bilateral injuries.
Furthermore, the procedures involved in extracting and culturing limbal cells are complex and resource-intensive. Ongoing research strives to refine these processes, aiming to make this innovative therapy more accessible and widely available, thereby mitigating the limitations currently faced by patients.
Patient Outcomes and Efficacy of CALEC
Patient outcomes from CALEC therapy have shown remarkable promise, with over 90 percent of participants experiencing significant improvements in their corneal condition. The therapy not only restored the corneal surface in many cases, but also enhanced visual acuity, allowing patients to regain functional sight after years of debilitating injuries.
The long-term follow-up results from the 18-month study indicate not only the efficacy of CALEC but also its safety, with minimal adverse events recorded. This sets a solid foundation for future endeavors aimed at expanding the clinical applications of stem cell technology in ophthalmology.
The Role of Research and Collaboration in Ocular Innovations
Collaborative research efforts have been fundamental in bringing CALEC therapy to fruition. Partnerships between Mass Eye and Ear, Dana-Farber Cancer Institute, and Boston Children’s Hospital have facilitated the necessary innovations and clinical trials to advance this treatment. Such interdisciplinary collaboration fosters a more comprehensive understanding of complex ocular conditions and drives the development of novel therapeutic strategies.
As discovery in ocular health progresses, the pooling of resources and expertise is essential. This not only allows for more rigorous testing of stem cell therapies but also ensures that findings are disseminated and utilized effectively across diverse medical settings, ultimately optimizing patient care.
Clinical Implications of CALEC Therapy
The clinical implications of CALEC therapy resonate beyond just the immediate treatment of corneal injuries; they pave the way for broader applications of stem cell therapies within ophthalmology. By demonstrating that cultivation and transplantation of limbal epithelial cells can successfully restore corneal damage, CALEC sets a precedent for similar strategies that could address other ocular diseases.
This expansion of therapeutic horizons presents a significant opportunity for ophthalmologists to adopt more innovative approaches to patient care. With promising research outcomes, CALEC therapy could soon become a standard part of treatment regimens for conditions that currently pose substantial challenges to visual restoration.
The Importance of Regulatory Approval in Stem Cell Therapies
Navigating the regulatory landscape is crucial for the advancement of therapies like CALEC. The approval process ensures that clinical applications of stem cell treatments adhere to safety and efficacy standards. As CALEC advances toward potential FDA approval, the research team emphasizes the need for thorough and ethically conducted trials to support their claims.
Regulatory scrutiny not only protects patients but also facilitates trust in emerging therapies. By adhering to rigorous standards, developers of CALEC can confirm the scientific validity of their approach, ultimately leading to wider acceptance and implementation of these groundbreaking treatments in clinical settings.
The Future of Ocular Regeneration Beyond CALEC
The promising results of CALEC therapy signify a shift towards a more regenerative approach to treating ocular conditions. As research continues to evolve, future therapies could build on the principles established by CALEC, exploring new avenues to regenerate and repair corneal and ocular tissue.
Looking ahead, the potential for integrating various forms of stem cell research, including advancements in bioengineering and cellular therapy, could lead to transformative treatments for a range of eye conditions. This holds the promise of not only restoring vision to those with corneal injuries but also addressing broader ocular health concerns effectively.
Frequently Asked Questions
What is therapie CALEC and how does it relate to stem cell therapy?
Therapie CALEC, or cultivated autologous limbal epithelial cells, is a groundbreaking treatment developed at Mass Eye and Ear that employs stem cell therapy to repair damaged corneal surfaces. This innovative procedure harvests healthy limbal epithelial stem cells from a patient’s unaffected eye, cultivates them, and then transplants them into the injured eye, promoting regeneration and restoring vision.
How does CALEC therapy assist in corneal repair for patients with ocular injuries?
CALEC therapy provides a new hope for patients suffering from ocular injuries that lead to corneal damage. By utilizing stem cells from a healthy eye, it can effectively regenerate the corneal surface, addressing the deficiencies caused by past injuries that were once considered untreatable.
Who will benefit from stem cell therapy like CALEC at Mass Eye and Ear?
Patients with corneal injuries, such as chemical burns or infections, who have experienced significant limbal stem cell deficiency are the primary candidates for CALEC therapy at Mass Eye and Ear. This treatment offers a promising option for those who have not responded to conventional corneal transplant surgeries.
What has research shown about the effectiveness of CALEC in clinical trials?
Recent clinical trials have demonstrated that CALEC therapy boasts over 90% effectiveness in restoring corneal surfaces, with significant improvements in vision reported by participants at both the 12-month and 18-month follow-ups, indicating its potential as a reliable treatment option.
What makes CALEC therapy a safer option for corneal repair compared to traditional methods?
CALEC therapy is considered safer than traditional corneal transplants because it uses a patient’s own stem cells, minimizing the risk of rejection or complications associated with foreign tissue. Additionally, major adverse events were rare, showcasing a high safety profile during trials.
Is CALEC therapy currently available for general use in hospitals?
Currently, CALEC therapy remains experimental and is not widely available in hospitals, including Mass Eye and Ear. Further studies are needed before it can receive federal approval and become accessible to a broader patient population.
What advances in ocular treatments does CALEC represent in the field of ophthalmology?
CALEC represents a significant advancement in ophthalmology, marking the first approved human clinical trial of a stem cell therapy specifically for corneal repair in the U.S., highlighting the potential of stem cell therapies to revolutionize treatment for otherwise untreatable ocular conditions.
How long does the CALEC procedure take from initial biopsy to transplantation?
The overall process for CALEC treatment takes approximately two to three weeks, including time for the initial biopsy of healthy limbal stem cells, cultivation, and preparation of the graft before the surgical transplantation into the damaged eye.
| Key Point | Description |
|---|---|
| Introduction of CALEC | Ula Jurkunas performed the first CALEC surgery at Mass Eye and Ear. |
| Stem Cell Therapy | A clinical trial using stem cells from a healthy eye restored corneal surfaces safely in 14 patients over 18 months. |
| Treatment Process | The CALEC involves harvesting stem cells, culturing them, and transplanting them into the damaged eye. |
| Effectiveness | The trial showed over 90% effectiveness in restoring corneal surfaces. |
| Patient Outcomes | 50% of participants fully restored vision after 3 months, increasing to 93% success rate over 12 months. |
| Safety Profile | No severe adverse events noted; one bacterial infection due to contact lenses. |
| Experimental Status | Currently not offered in the U.S. yet; requires more studies for FDA approval. |
| Future Research | Future studies aim to include more patients across multiple centers for better data. |
Summary
Therapie CALEC is pioneering a new frontier in treating corneal damage with an impressive success rate of over 90%. This innovative stem cell therapy not only restores the surface of the eye but offers renewed hope for patients previously deemed untreatable. With ongoing research and future trials aiming to expand its availability, CALEC stands to revolutionize ocular care as we know it.